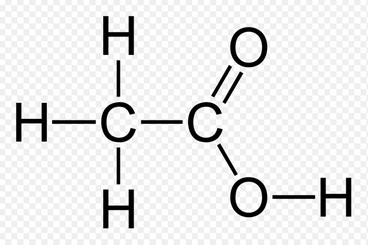
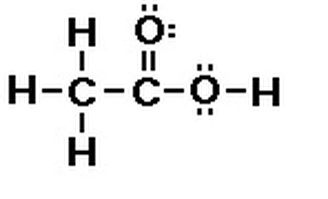Atomic StructureIn this image, you would notice that there are lines connecting between the elements Hydrogen, Oxygen and Carbon. Those lines are what represent the bonds between each element. The singular lines are the singular bonds and the double lines are the double bonds. In the singular bonds, there are two electrons, making each outer shell orbit of the atoms to become complete. This means that the compound is a covalent bond.
|
What is Vinegar?
Vinegar is a liquid substance used today in various ways. One of the main ways people used vinegar back then and today is for flavoring recipes. Vinegar could be used in such recipes like flavoring your salad. It adds a sour taste to balance such a plain meal. Adding vinegar to recipes like salad, tend to make the meal more appetizing and satisfying to eat. |
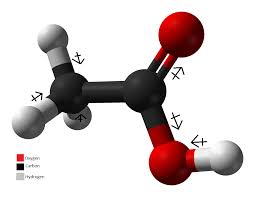
Molecular StructureRecall that the chemical formula for acetic acid is CH3COOH. Knowing this, you will find that Figure 3 shows an image of the molecular structure for acetic acid. As you can see, the red spheres represent Oxygen, black spheres represent Carbon, and white spheres represent Hydrogen. For the Oxygen and Carbon elements, there are 2 of each. Whereas there are 4 elements of Hydrogen. This makes acetic acid a carbon and acidic compound.
|
KEY WORDS:
- Characteristic Properties: A characteristic property is a chemical or physical property that helps identify and
classify substances. The characteristic
properties of a substance are always the same whether the sample
one is observing is large or small.
- Boiling Point: the temperature at which a liquid boils and turns to vapor.
- Melting Point: the temperature at which a given solid will melt.
- Density:
- -the degree of compactness of a substance.
- -degree of consistency measured by the quantity of mass per unit volume.
- Viscosity: the ability for a substance to flow, the state of being thick, sticky, and semifluid in consistency, due to internal friction.
- Covalent Bond: A covalent bond is
a chemical bond that
involves the sharing of electron pairs between atoms. The stable balance of
attractive and repulsive forces between atoms when they share electrons is
known as covalent bonding.
- Single Bond: a chemical bond in which one pair of electrons is shared between two atoms.
- Double Bond: a chemical bond in which two pairs of electrons are shared between two atoms.
- Miscibility: the property of substances to mix in all proportions, forming a homogeneous solution.
- Solubility: the ability for a given substance, the solute, to dissolve in a solvent.
- Acidity: the level of acid in substances such as water, soil, or wine.
- pH: The pH of a solution is a measure of the molar concentration of hydrogen ions in the solution and as such is a measure of the acidity or basicity of the solution. The letters pH stand for "power of hydrogen" and numerical value for pH is just the negative of the power of 10 of the molar concentration of H+ ions.
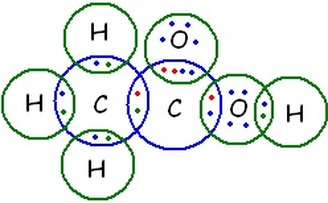
Bohr Rutherford Diagram for Compound This is an image explaining more clearly of how the elements are a covalent bond, and shows how each electron is shared with another. Recall that acetic acid is made up of the elements; Carbon, Hydrogen, and Oxygen. As you see in the picture, there are single bonds that connect the Carbon to the rest of the formula, and to the Hydroxide within the structure. There is also a double bond between the Oxygen and one of the Carbon atoms. You can identify this by looking at how many pairs there are within each shared electron. If you count the electrons, you will find that there are 24 valence electrons in total. This diagram could be transformed into a Lewis Dot Structure which will show the singular and double bonds with lines.
|
Lewis Dot StructureHere is a diagram of the Lewis Dot Structure for acetic acid. It is quite similar to the atomic structure, except it shows the electrons. This is how it would look when the compound is drawn in Lewis Dot structure purposely to show the specific bonds within the diagram. Taking a look back at the Bohr Rutherford Diagram for the compound, you will notice how there are two electrons making the two elements have a complete shell, this means that there are two electrons shared for each line in the Lewis Dot structure. If you count each line representing pairs of electrons including the electrons that were not shared, you will again, identify that there are 24 valence electrons.
|
Physical Properties
|
Chemical Properties
|
|
|
How does Vinegar react with Baking Soda?
|